
The quantity of charge associated with the constituent ions and the spacing between the ions are the two fundamental factors that influence the lattice energy of an ionic compound. NCERT Exemplar Class 12 Chemistry solutions Chapter 1 The Solid State.NCERT solutions for Class 12 Chemistry Chapter 1 The Solid State.The volume change is denoted by V m (per mole).Īs a result, while computing the lattice energies of ionic solids, the outside pressure is also taken into account. The molar lattice enthalpy is denoted by Δ GH. The molar lattice energy is denoted by Δ GU. The following equation can be used to represent the molar lattice energy of an ionic crystal in terms of molar lattice enthalpy, pressure, and volume change: Difference Between Lattice Energy and Lattice Enthalpy Although lattice energy cannot be measured empirically, it can be calculated or inferred using electrostatics or the Born-Haber cycle. The energy required to split a mole of an ionic solid into gaseous ions is known as lattice energy. Lattice energy must always have a negative value, according to this definition. Some sources define lattice energy in the opposite way, as the amount of energy released during the exothermic formation of an ionic solid from its gaseous ionic ingredients. As a result, the value of this quantity is always positive. It can also be described as the amount of energy required to split one mole of an ionic crystal into gaseous ions in a vacuum via an endothermic process. The energy required to transform one mole of an ionic solid into gaseous ionic components is known as lattice energy. This amount is usually given in kilojoules per mole (kJ/mol). The Born-Haber cycle, on the other hand, can be used to estimate it. An ionic solid's lattice energy cannot be directly measured. It reveals information on ionic materials' volatility, solubility, and hardness, among other things. MgO the higher charge on Mg leads to a larger lattice energyĦ.The strength of the ionic bonds in an ionic substance is measured by lattice energy.Li 2O the higher charge on O 2– leads to a larger energy additionally, Cl – is larger than O 2– this leads to a larger interionic distance in LiCl and a lower lattice energy.MgO the higher charges on Mg and O, given the similar radii of the ions, leads to a larger lattice energy.MgO selenium has larger radius than oxygen and, therefore, a larger interionic distance and thus, a larger smaller lattice energy than MgO.The compounds with the larger lattice energy are Since the lattice energy is negative in the Born-Haber cycle, this would lead to a more exothermic reaction.Ĥ. 4008 kJ/mol both ions in MgO have twice the charge of the ions in LiF the bond length is very similar and both have the same structure a quadrupling of the energy is expected based on the equation for lattice energyĥ. The smaller the radius of the anion, the shorter the interionic distance and the greater the lattice energy would be.In the Born-Haber cycle, the more negative the electron affinity, the more exothermic the overall reaction. A higher electron affinity is more negative.The lower it is, the more exothermic the reaction will be. 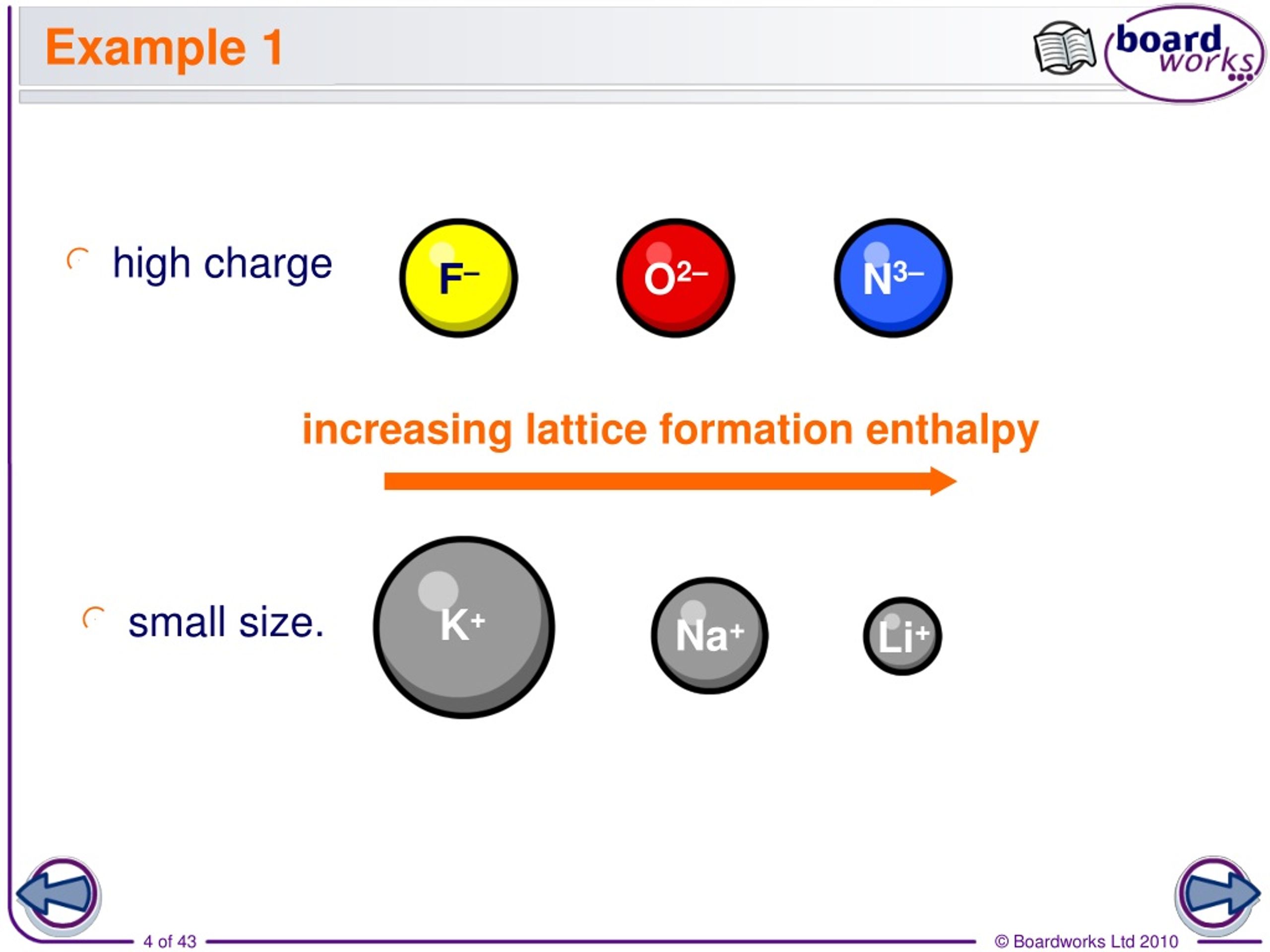
As in part (b), the bond energy is a positive energy.This would make the reaction more exothermic, as a smaller positive value is “more exothermic.” A lower ionization energy is a lower positive energy in the Born-Haber cycle.Since the lattice energy is negative in the Born-Haber cycle, this would lead to a more exothermic reaction. The smaller the radius of the cation, the shorter the interionic distance and the greater the lattice energy would be.Recall that the more negative the overall value, the more exothermic the reaction is. In each case, think about how it would affect the Born-Haber cycle. The answer is (d), which requires about 740 kJ/mol.ģ. U may be calculated from the Born-Haber cycle. The lattice energy, U, is the energy required to convert the solid into separate ions. We begin with the elements in their most common states, Cs( s) and F 2( g). The Born-Haber cycle shows the relative energies of each step involved in the formation of an ionic solid from the necessary elements in their reference states.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
